Now playing video 3 of 4
Diamond and graphite
Description
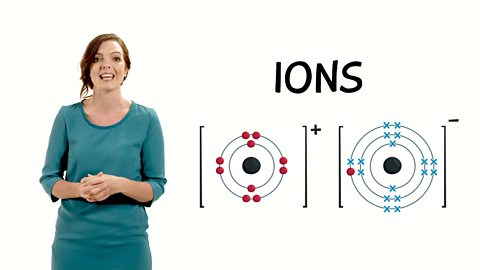
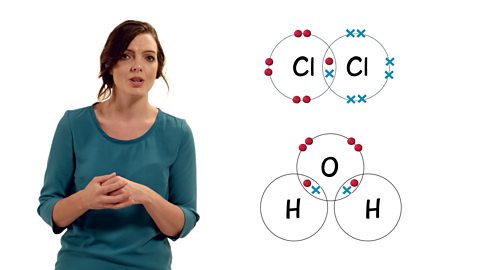
"This video explains why diamond and graphite have different properties depsite both being made of carbon. Diamond has a giant covalent structure with strong bonds, meaning that there are no free electrons. Graphite has one delocaslised electron which is free to move, and weak forces of attraction between the layers.
Find out more about GCSE Chemistry"
Now playing video 3 of 4
- 3:14
- 3:54
- Now playing3:02

- Up next2:16
